We are interested in the mechanisms underlying spontaneous activity in the developing nervous system and the role this activity plays in the construction of neuronal circuits. There are several examples throughout the developing vertebrate nervous system, including the retina, spinal cord, hippocampus and neocortex, where immature neural circuits generate activity patterns that are distinct from the functioning adult circuitry. It has been proposed that these transitional circuits provide the test patterns necessary for normal development of the adult nervous system. Here we study this phenomenon in the mouse retina. Click on the headings to learn more!
2-photon calcium imaging of spontaneous activity in retina isolated from early postnatal mice. Showing fractional change in fluorescence, 10x real time, 1 mm per side
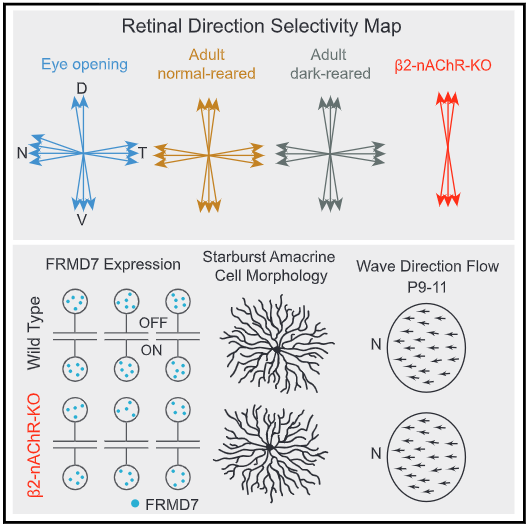
Summary data from Tiriac et al ,2022 showing the development of horizontal direction selectivity is dependent the presence of cholinergic retinal waves
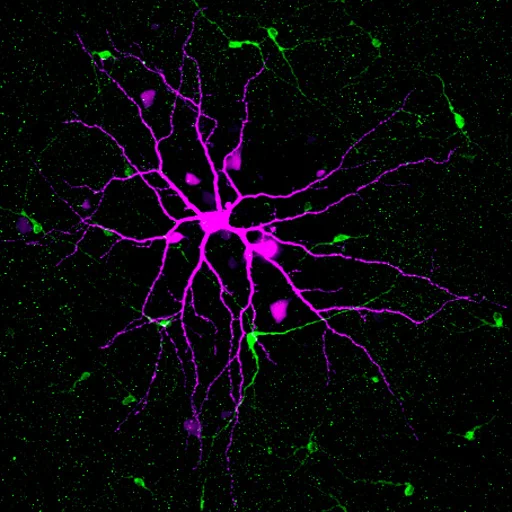
We have several studies showing how retinal wave interact with ipRGCs during development
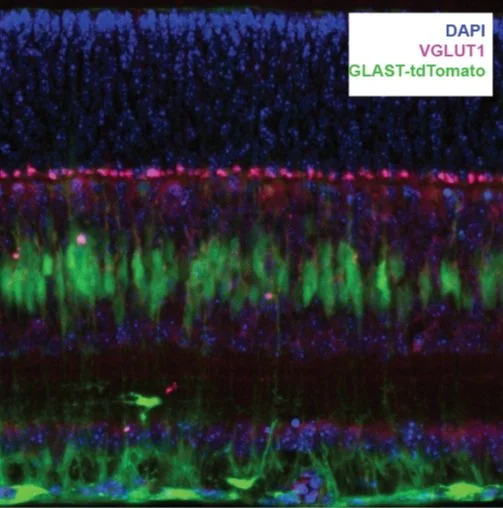
Current projects test a role for neuronal glial signaling during retinal waves.